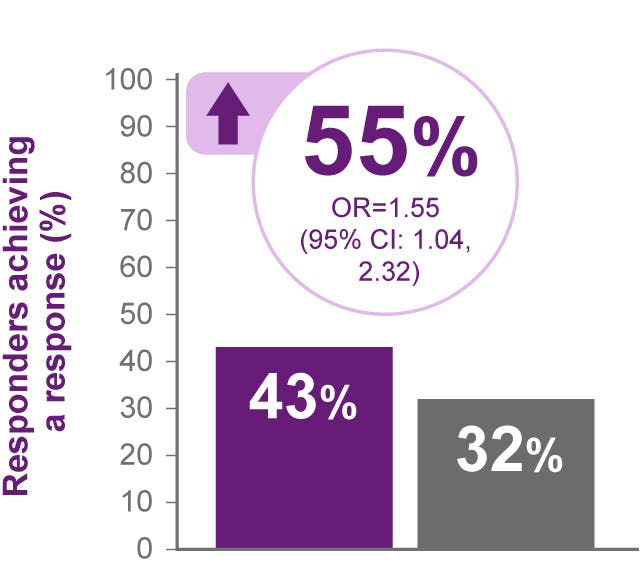
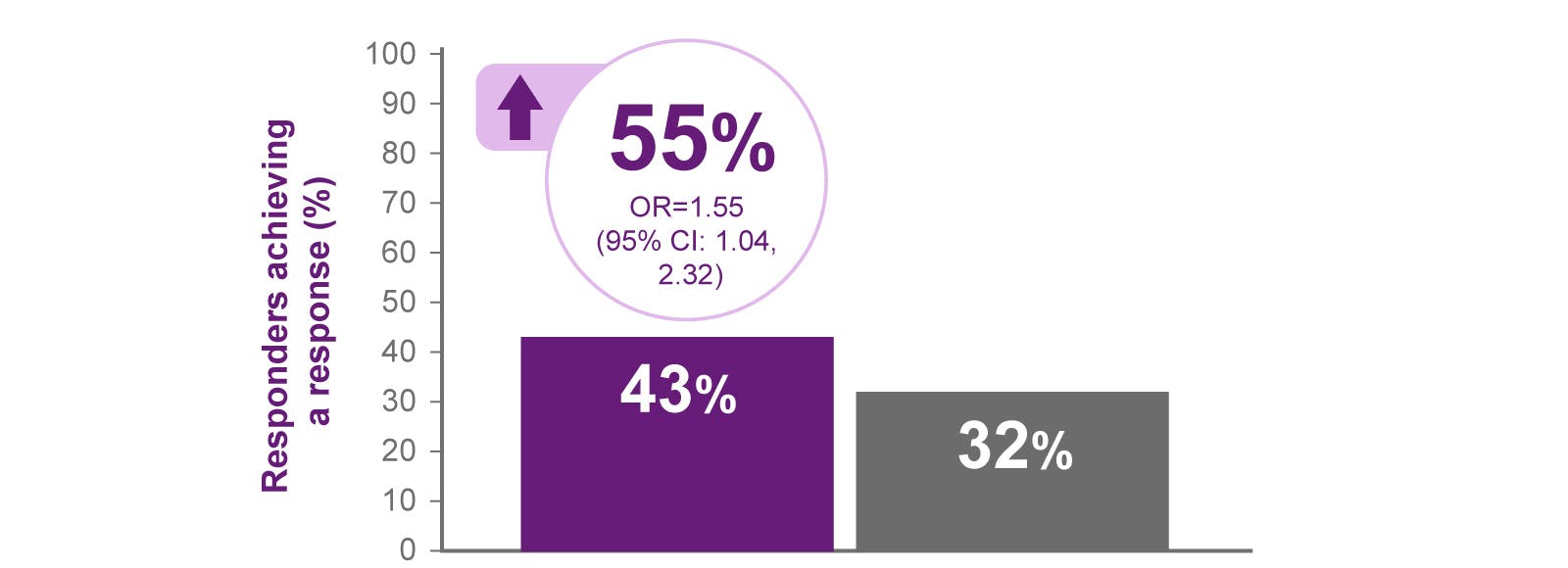
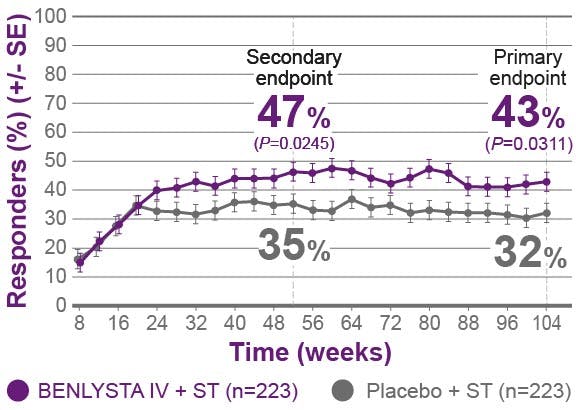
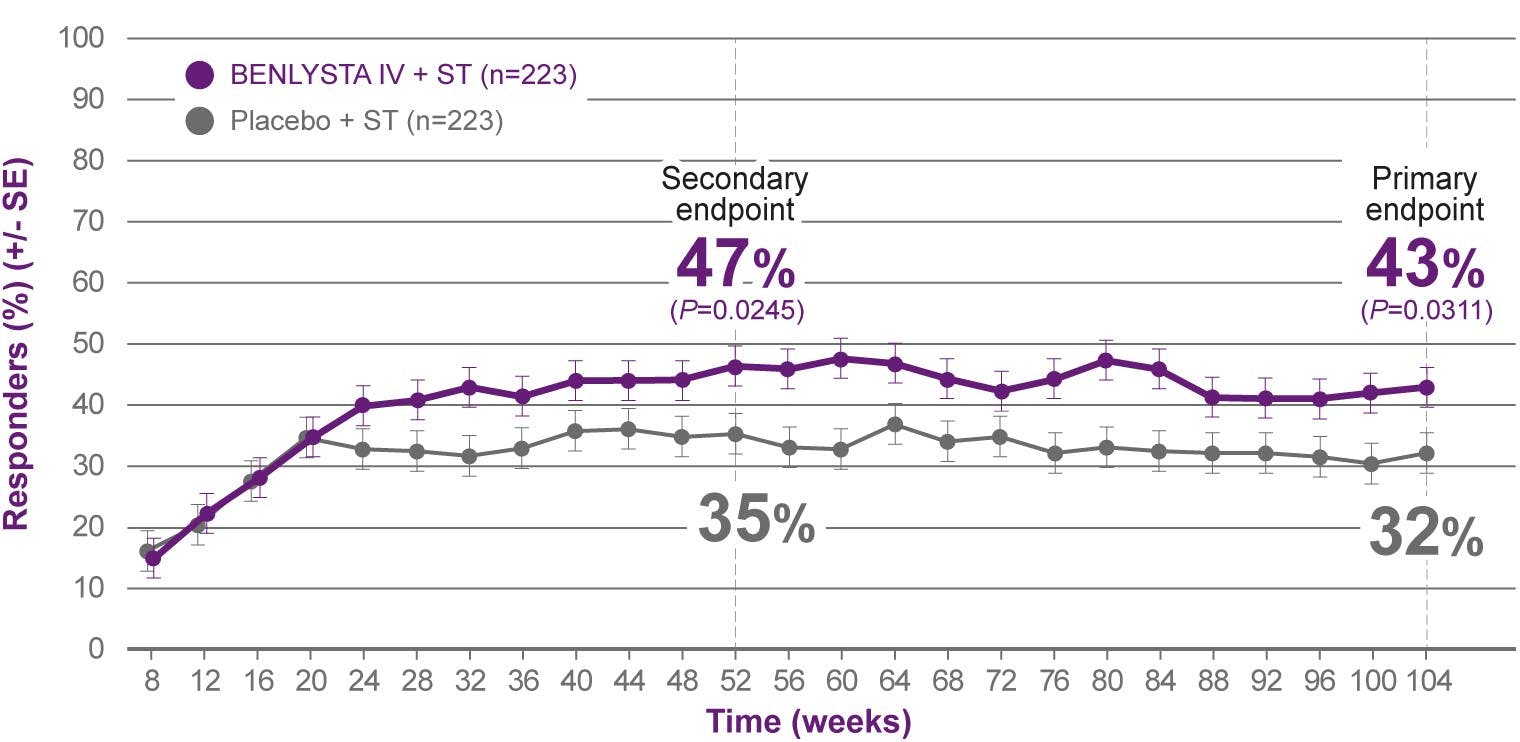
BENLYSTA is indicated for patients aged ≥5 with active systemic lupus erythematosus (SLE) or active lupus nephritis who are receiving standard therapy. BENLYSTA is not recommended in patients with severe active central nervous system lupus.
BENLYSTA is indicated for patients aged ≥5 with active systemic lupus erythematosus (SLE) or active lupus nephritis who are receiving standard therapy. BENLYSTA is not recommended in patients with severe active central nervous system lupus.
BENLYSTA is indicated for patients aged ≥5 with active systemic lupus erythematosus (SLE) or active lupus nephritis who are receiving standard therapy. BENLYSTA is not recommended in patients with severe active central nervous system lupus.
CONTRAINDICATION
Previous anaphylaxis with BENLYSTA.
CONTRAINDICATION
Previous anaphylaxis with BENLYSTA.
WARNINGS AND PRECAUTIONS
Serious Infections: Serious and sometimes fatal infections have been reported and occurred more frequently with BENLYSTA.
CONTRAINDICATION
Previous anaphylaxis with BENLYSTA.
WARNINGS AND PRECAUTIONS
Serious Infections: Serious and sometimes fatal infections have been reported and occurred more frequently with BENLYSTA. Use caution in patients with severe or chronic infections, and consider interrupting therapy in patients with a new infection.
Progressive Multifocal Leukoencephalopathy (PML): Cases of JC virus-associated PML resulting in neurological deficits, including fatal cases, have been reported. If PML is suspected, immunosuppressant therapy, including BENLYSTA, must be suspended until PML is excluded. If confirmed, stop immunosuppressant therapy, including BENLYSTA.
Hypersensitivity Reactions (Including Anaphylaxis): Acute hypersensitivity reactions, including anaphylaxis and death, and infusion-related reactions have been reported. Generally, reactions occurred within hours of the infusion but may occur later, including in patients who have previously tolerated BENLYSTA. Non-acute hypersensitivity reactions (eg, rash, nausea, fatigue, myalgia, headache, and facial edema) typically occurred up to a week after infusion. Monitor patients during and after treatment and be prepared to manage anaphylaxis and infusion-related reactions. Be aware of the risk of hypersensitivity reactions, which may present as infusion-related reactions. Discontinue immediately in the event of a serious reaction. With intravenous administration, if an infusion reaction develops, slow or interrupt the infusion.
Depression and Suicidality: Depression and suicidality were reported in patients receiving BENLYSTA. Before adding BENLYSTA, assess patients’ risk of depression and suicide and monitor them during treatment. Instruct patients/caregivers to contact their HCP if they experience new/worsening depression, suicidal thoughts/behavior, or other mood changes.
Malignancy: There is an increased risk of malignancies with the use of immunosuppressants. The impact of BENLYSTA on the development of malignancies is unknown.
Immunization: Live vaccines should not be given for 30 days before or concurrently with BENLYSTA as clinical safety has not been established.
Use With Biologic Therapies: Available data do not support the safety and efficacy of concomitant use of BENLYSTA with rituximab in patients with SLE. An increased incidence of serious infections and post-injection systemic reactions in patients receiving BENLYSTA concomitantly with rituximab compared to patients receiving BENLYSTA alone has been observed. The safety and efficacy of BENLYSTA concomitantly with other biologic therapies, including B-cell-targeted therapies, have not been established. Caution should be exercised if BENLYSTA is administered in combination with other biologic therapies.
ADVERSE REACTIONS
The most common serious adverse reactions in adult SLE clinical trials were serious infections; some were fatal. The most common adverse reactions (≥5%) were nausea, diarrhea, pyrexia, nasopharyngitis, bronchitis, insomnia, pain in extremity, depression, migraine, pharyngitis, and injection site reactions (subcutaneous injection).
Adverse reactions reported in clinical trials with SLE pediatric patients (≥5 years) and adult patients with lupus nephritis were consistent with those observed in adult SLE trials.
USE IN SPECIFIC POPULATIONS
Pregnancy: There are insufficient data in pregnant women to establish whether there is drug-associated risk for major birth defects or miscarriage. After a risk/benefit assessment, if prevention is warranted, women of childbearing potential should use contraception during treatment and for ≥4 months after the final treatment.
Pregnancy Registry: HCPs are encouraged to refer patients and pregnant women are encouraged to enroll themselves by calling 1-877-311-8972 or visiting https://mothertobaby.org/ongoing-study/benlysta-belimumab/.
Please see full Prescribing Information and Medication Guide for BENLYSTA.